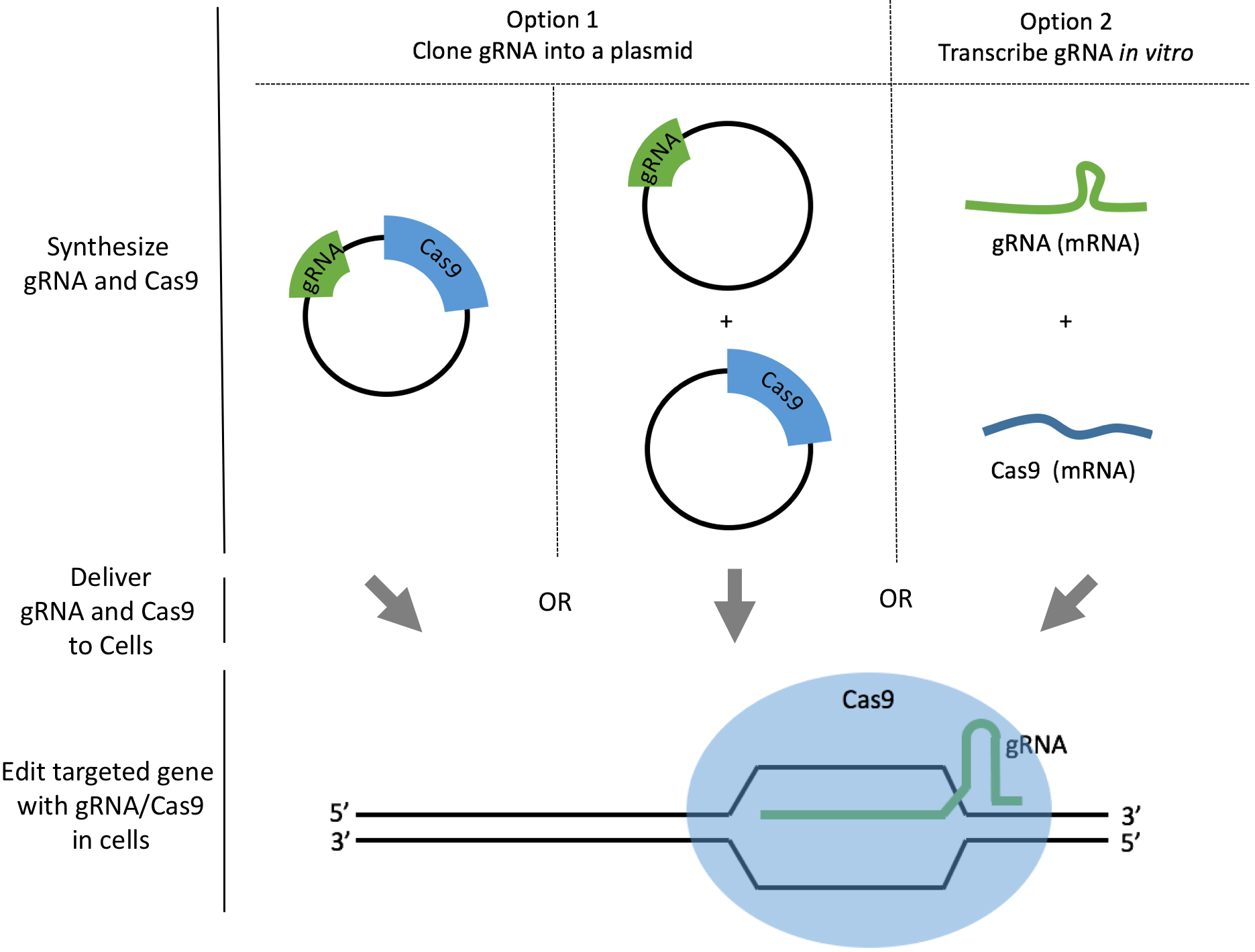
How to Design gRNAs to Target Your Favorite Gene
This tutorial is part 2 of 4 in the series “How to Create Knockouts Using CRISPR.” Use Benchling's free molecular biology tools to plan your own CRISPR experiment and design your own gRNAs here.
OK. Now we have decided the cell line and Cas9 for our experiments. Time to start designing gRNAs!To eliminate the function of your favorite gene, you want to target a consecutively-expressed exon near the 5’ end. This design has a high likelihood of creating a nonfunctional protein when frameshift mutations happen. Here we will walk through all the necessary steps, and discuss important factors to consider in your gRNA design. We will also show how you can design gRNAs using Benchling.
Step 1. Identify the genomic region you want to target.
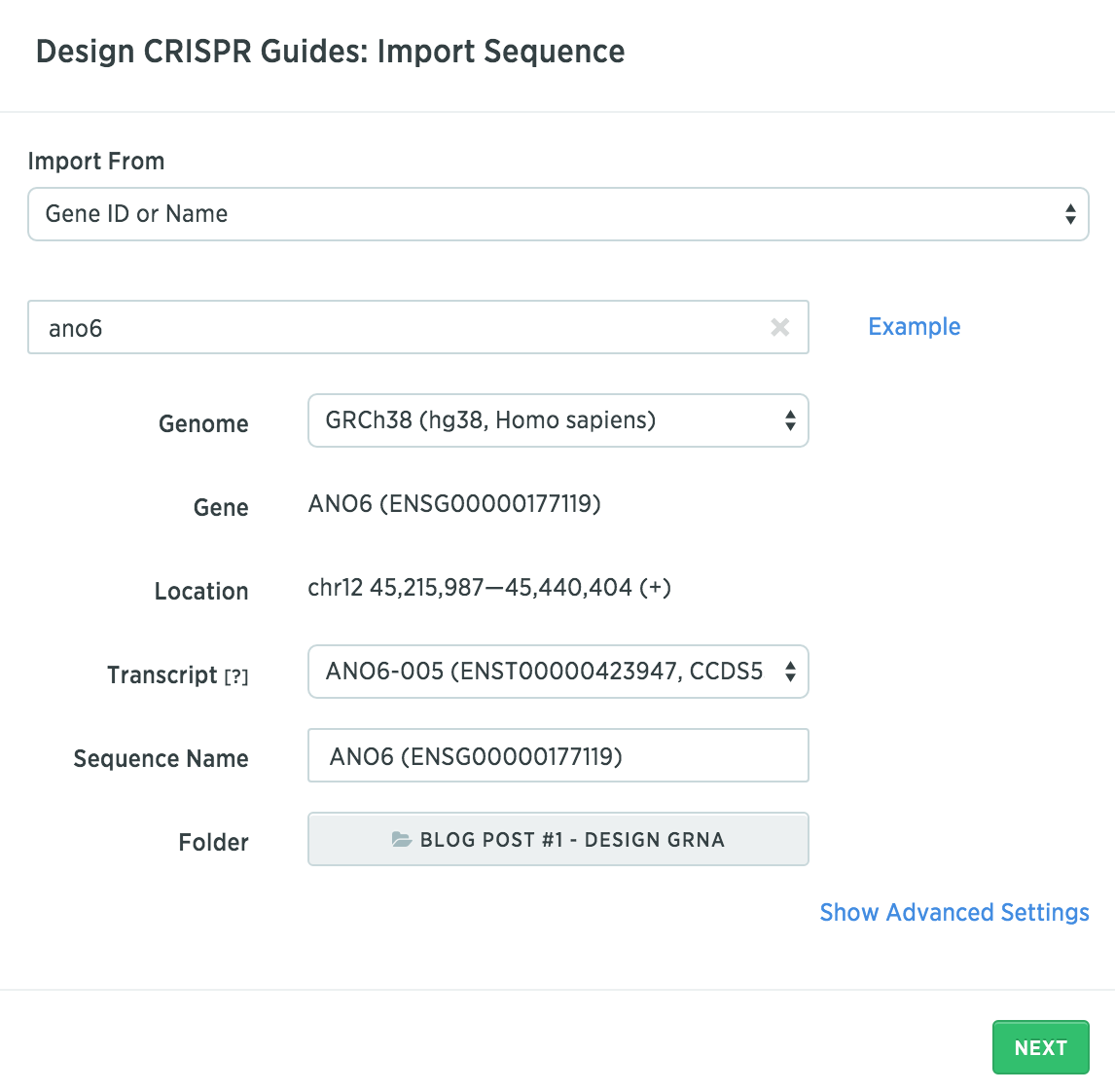
You need to first find which exons are present in all transcript variants of your targeted gene. To get this information in Benchling, start by clicking “Create” in the top right and then click “Create CRISPR”. In the next dialog box, you can search for your gene of interest. Here is what it looks like to search for Ano6 in a human genome (hg38):
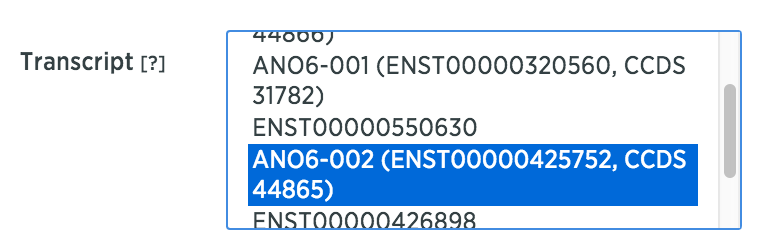
In Advanced Settings, you can select all transcripts to apply the annotations on the genome.
Then you can view the exons of all transcript variants of your gene of interest:
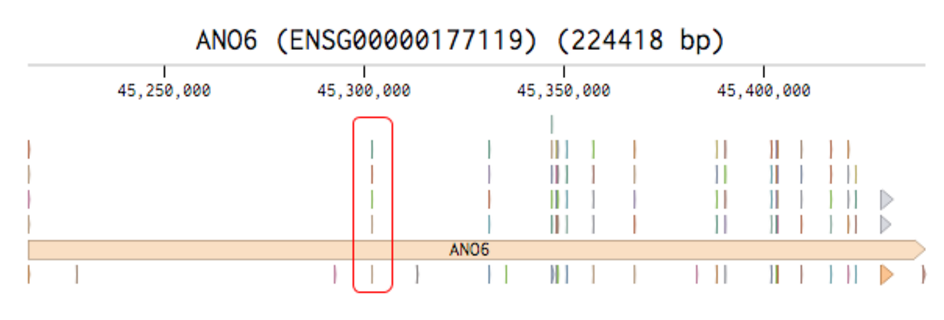
To make Ano6 knockout cells using CRISPR, you could design gRNAs to target the exon in the red rectangle, because it is included in all Ano6 isoforms and close to the 5’ end.
Tip
Make sure there are no critical functional domains upstream of your target site because the resulting truncated protein may still have some activity. You can analyze protein functional domains in InterPro or MOTIF.
Step 2. Find all possible protospacer sequences around your intended target site.
Now, we will show you how to find protospacer sequences directly upstream of the PAM sequence for your chosen Cas9, again using Benchling as an example.
Click the exon you want to target on the sequence map to select it, and then add it as a target region in the Design CRISPR tab.
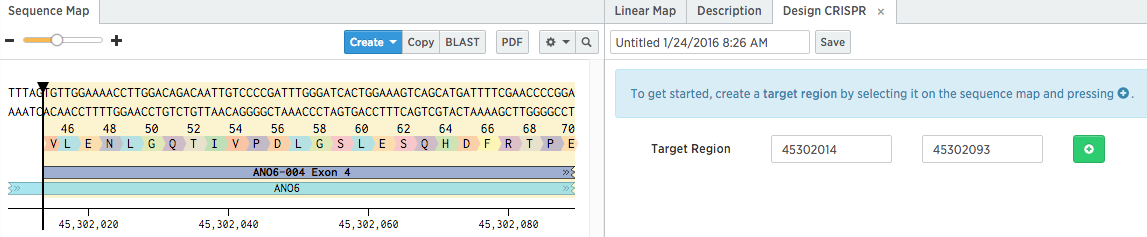
Tips
As Fu et al.showed, a truncated gRNA with 17 or 18 nucleotides can decrease off-target effects without compromising the cleavage efficiency [1]. It might be worth trying a truncated protospacer to increase the specificity of your experiments.
You could also reduce off-target effects by using paired Cas9 nickases to induce indels [2] [3].
Step 3. Select at least two protospacer sequences that minimize off-target effects
Sometimes, one gRNA works better than another. Given this, we would recommend that you design and test more than two protospacer sequences to increase the success rate of creating gene knockouts.
Benchling calculates on-target and off-target scores for every protospacer sequence. The scores are calculated based on algorithms developed by Doench et al. [4] and Hsu et al. [5] Both scores are between 0 to 100, with higher scores being better. These two scores can guide you to select good protospacer sequences.
The off-target score tells you the inverse probability of Cas9 off-target binding. A higher score means the sequence has less chance to bind to sequences in the rest of the genome.
The on-target score represents the cleavage efficiency of Cas9 [6]. You can think of the score as the probability a given gRNA will be in top 20% of cleavage activity. Note that the scoring system is not linear, and only 5% of gRNAs receive a score of 60 or higher. Recently, Doench et al. improved algorithms for predicting on-target and off-target activities[7]. Benchling will modify the scoring algorithm to stay up-to-date.
Tips
Since Cas9 will cut both DNA strands after binding to the target sequence, your protospacer sequence can be on either the plus or minus strand.
You might want to examine potential off-target sequences to determine whether these sequences are part of the genes relevant in your studies. In Benchling you can click on the off-target score to see these sequences (mismatches are denoted in red):
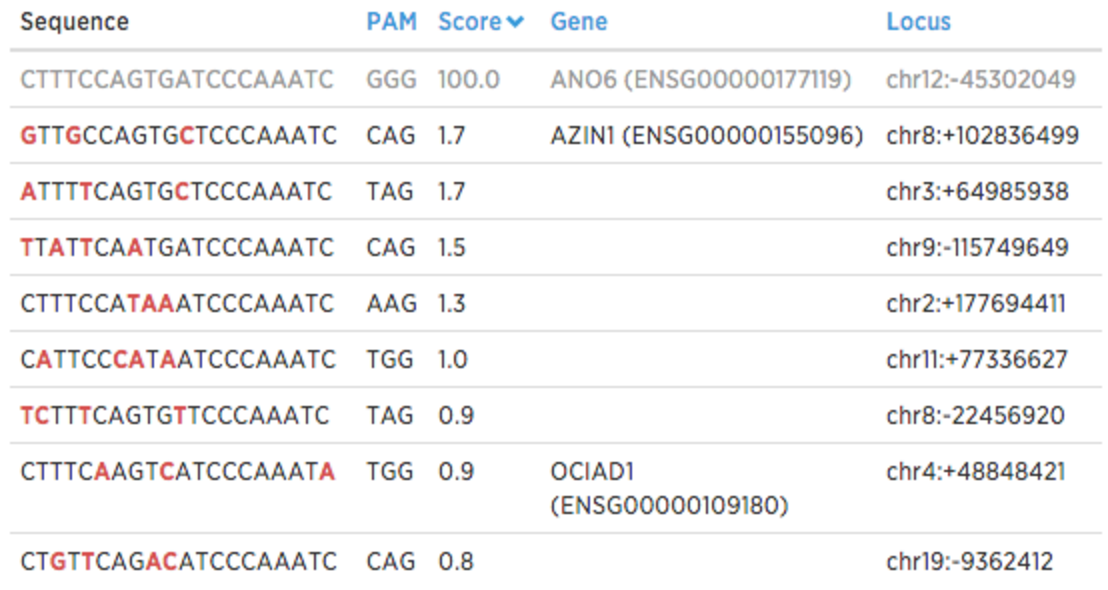
What’s next?
Now you have selected at least two gRNAs to target your favorite gene. It’s time to bring the digital designs to the bench! In our next blog post, we will talk about how to clone your gRNAs to vectors of your choice. After that, we will discuss CRISPR delivery strategy, assays to detect indels and how you select gene knockouts. Follow us on Twitter or Facebook so you don’t miss the next post.
Want to use Benchling's CRISPR tools at your company?
References
Fu, Yanfang et al."Improving CRISPR-Cas nuclease specificity using truncated guide RNAs." Nature biotechnology 32.3 (2014): 279-284.
Ran, F Ann et al."Double nicking by RNA-guided CRISPR Cas9 for enhanced genome editing specificity." Cell 154.6 (2013): 1380-1389.
Ran, F Ann et al."Genome engineering using the CRISPR-Cas9 system." Nature protocols 8.11 (2013): 2281-2308.
Doench, John G et al."Rational design of highly active sgRNAs for CRISPR-Cas9-mediated gene inactivation." Nature biotechnology (2014).
Hsu, Patrick D et al."DNA targeting specificity of RNA-guided Cas9 nucleases." Nature biotechnology31.9 (2013): 827-832.
Shalem, Ophir et al. "Genome-scale CRISPR-Cas9 knockout screening in human cells." Science 343.6166 (2014): 84-87.
Doench, John G et al. "Optimized sgRNA design to maximize activity and minimize off-target effects of CRISPR-Cas9." Nature Biotechnology(2016).