Introducing Benchling Validated Cloud: Accelerating Product Development for Life Sciences
Benchling is excited to introduce Benchling Validated Cloud, making it possible for Research and Development teams to work together from a unified, validated informatics solution. Just as those working in Research leverage Benchling for molecular design, experimentation, and sample registration, those working in early and late stage Development can access the same easy-to-use data management capabilities now within a controlled, compliant environment. Benchling's unified offering enables Research and Development teams to collaborate across a harmonized data model with the same structured, queryable data. With Benchling Validated Cloud, R&D teams will be able advance programs to clinical development more effectively as a result.
Reduce the Friction Between Phases of Research and Development

Many life science companies struggle to advance programs from scientific discovery to product development and manufacturing. Typically this handoff entails different processes, objectives, data management solutions, and teams unfamiliar with working together which leads to delay or incomplete knowledge transfer. Additionally, whereas scientific discovery teams prefer flexible data systems, clinical teams require control, repeatability, and scalability for high-quality development and manufacturing. The development and manufacturing phases are further complicated by more stringent regulatory requirements. Benchling's introduction of Validated Cloud offers a way to unify research and development teams on a single, compliant software solution in order to improve the tech transfer process so products can move efficiently into and through development.
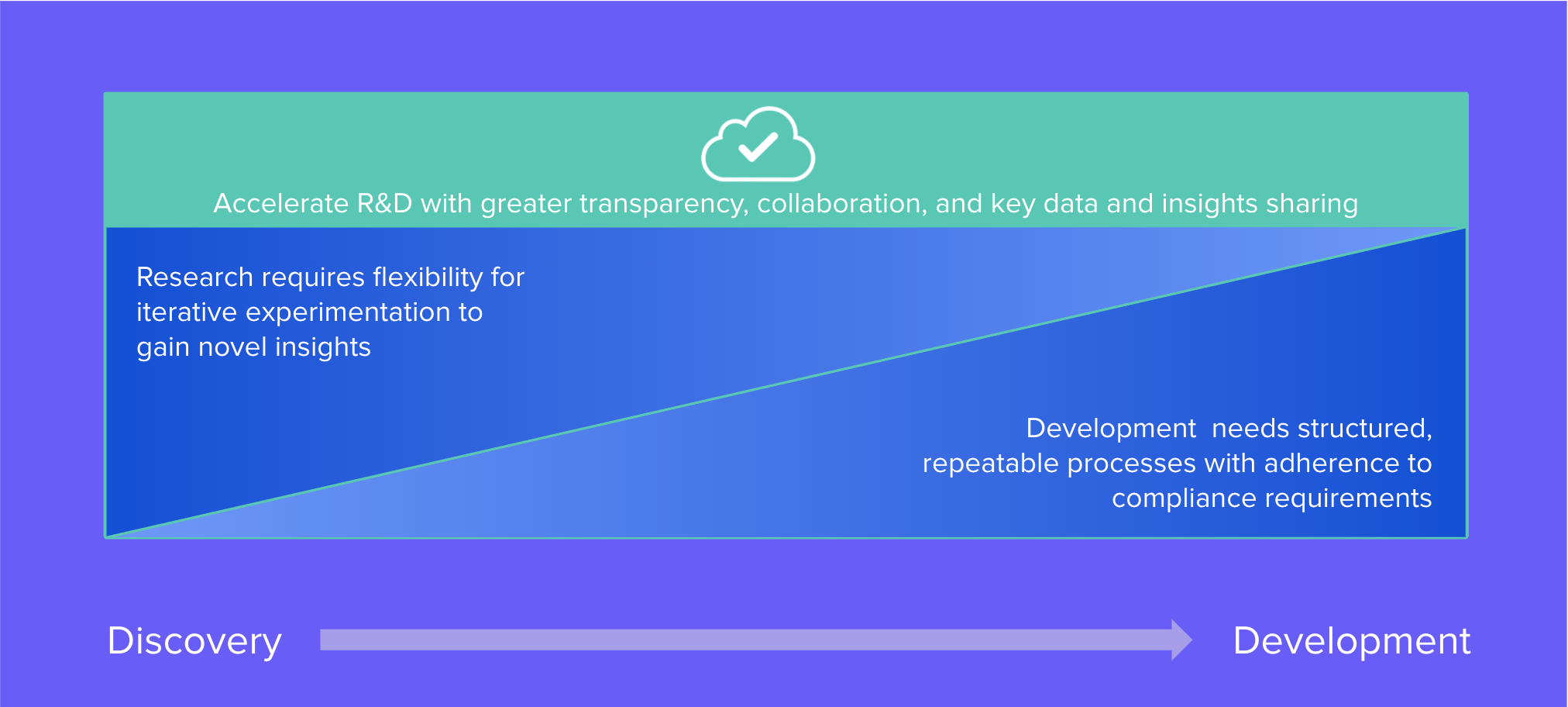
Benchling can play a vital role in reducing data silos and associated onerous work involved with the tech transfer between phases. To streamline this collaboration, Benchling Validated Cloud supports the controlled and repeatable work done in development within validated, compliant environments. R&D teams can maintain the same approach to managing biological registration, data collection, and structured insights. They can coordinate more closely with shared visibility to critical operational resources, program management, and scientific insights. With all of this connectivity, we foresee that Benchling Validated Cloud will also make the assembly of data packaging for regulatory filings faster and easier.
Download the Product Datasheet
Comply with Validation and Regulatory Requirements
Teams focusing on pre-clinical and development work require software that adheres to GxP systems requirements. To support getting the product to market, Benchling Validated Cloud conforms to standard validation and compliance practices by offering GxP-guided features, processes, and services. These capabilities include:
Dedicated Tenant and New Features
Validated Cloud provides a separate, dedicated tenant for validated applications that allows for greater control and compliance without impacting work done in non-validated tenant environments. New features in Benchling focus primarily on application controls to ensure user compliance per 21 CFR Part 11 and Annex 11 regulations, in the U.S. and E.U. respectively. These include electronic signature control on all entries, with records becoming locked and non-editable upon signature, and ability to track and trace engagement and changes within the system. The audit trail provides a summary of access with strict versioning of all edits and visible timestamps.
Quarterly Releases and Validation Support
Software updates within Validated Cloud tenants are released with controlled, quarterly cycles. With each release, customers receive Validation Plan and Impact Assessments to help determine the risk level of changes. Benchling’s Customer Success teams also provide supporting documents to facilitate the validation process (IQ, OQ, PQ, and UAT). Benchling’s Quality and Professional Services teams are also available to provide additional support to assist customers with how to best take advantage of Benchling capabilities across tenants throughout each organization’s Implementation, Validation, and Release and Change Management processes.
“Our research teams have been using Benchling for years, seen impressive results and are particularly reliant on the solution’s powerful collaboration capabilities. With Benchling Validated Cloud, we can now also meet regulatory compliance requirements and operate within the constraints of our rigid LIMS system.”Nermin Avdispahic, Quality Control Manager, New England Biolabs®
Learn More About Benchling Validated Cloud
If your R&D teams work in validated environments and might benefit from working on a unified informatics solution, please reach out.