Strengthening the biotech & tech connection: In conversation with Andreessen Horowitz’s Jay Rughani, Benchling’s Sajith Wickramasekara

Biopharma cares about getting cutting-edge medicines to market faster. They’re often asked to do more with less, especially in today’s resource-constrained economic climate. Integrating modern software, becoming cloud-first, and adopting a data-driven, digital approach are key to accelerating discovery and development. But what should these technical capabilities look like? How do they fit into the R&D lab ecosystem?
These were the main questions being asked at a recent, intimate event in Carlsbad, CA with leaders from the world’s top biopharma companies. And the intersection of technology and biotech was quite literally on center stage. Benchling’s Sajith Wickramasekara and Jay Rughani, investment partner at Andreessen Horowitz focused on Bio + Health, shared the stage to discuss trends in technology for the biotech industry, covering the themes that matter most to this audience — everything from data organization and integrity to hiring technical talent and the impact of AI on drug discovery. Below are some highlights from Sajith and Jay’s discussion.
On the forces increasing demand for SaaS in biotech
Jay: You have an interesting cross-sectional view into biotechnology R&D at Benchling, working with 1,000+ customers across multiple industries from biopharma to agriculture, consumer packaged goods, and more. What are the major challenges you’re hearing from scientists as it relates to the tools they need to do their best work?
Sajith: In the decade since we started Benchling, biotech has been innovating at a breakneck pace. The industry invented single-cell sequencing, brought the cost of DNA synthesis down to a few cents per base pair, and pioneered revolutionary gene editing systems ranging from CRISPR/Cas9 to base editing. We’ve seen so many industry firsts: the first cell therapy approved, the first in-human trial of a CRISPR gene editing medicine, and of course, the first dose of an mRNA vaccine.
Meanwhile, software for science hasn’t experienced the same step function change. Scientists are often held back by rigid, function-specific software and disconnected tools designed for compliance, not cutting-edge biology.
We hear three trends driving the need for new R&D IT capabilities:
Science is growing increasingly complex. Legacy software was not built for biological drugs like bispecific antibodies, RNA, or cell and gene therapies. What was once a simple molecule that could be drawn on a whiteboard and digitally represented with a string of text, is now a complex protein whose structure we don’t always know and takes a multi-dimensional data model to represent.
There’s an explosion of scientific data from new analytical techniques and lab automation. Labs are faced with exponential increases in the volume and complexity of the data they have to manage. Today that data lives in silos, scattered across multiple disconnected systems in an unstructured way. Labs lack the ability to easily integrate data and context at scale. They struggle to apply AI, ML, and analytics because they don’t have clean, well-organized, structured data or data systems.
Science is becoming increasingly specialized, requiring deeper collaboration within and outside organizations. With more collaboration, scientific workflows now require a significant number of hand-offs and better integration across a fragmented software ecosystem.
The reality is that these factors are impacting scientists’ productivity, data quality, and overall R&D timelines and efficiency. Digital transformation has so much potential in biotechnology.
Jay: R&D efficiency is the priority. How can biopharma companies harness the increasing advancements in biology to deliver more breakthrough medicines to patients, while the macroeconomic environment and the rising cost of capital increases budgetary pressures on these companies? In other words, how can they do more with less? They need to become software companies. Modern software has increased the throughput of so many other industries -- from banking, to media, to automotive; imagine what’s possible if scientists actually had access to the best software tools available?
The most-forward thinking biopharma executives I know are investing in digitally transforming their enterprises, while streamlining waste elsewhere.
On big opportunities ahead in biotech
Jay: My colleague Vijay Pande at Andreessen Horowitz predicted that AI will ultimately be more important for bio than in enterprise technology.
In biology, AI can be incredibly enabling: It helps turn tasks that used to be manually intensive, repetitive, and human error-prone into industrialized processes, freeing humans to focus on more meaningful work.
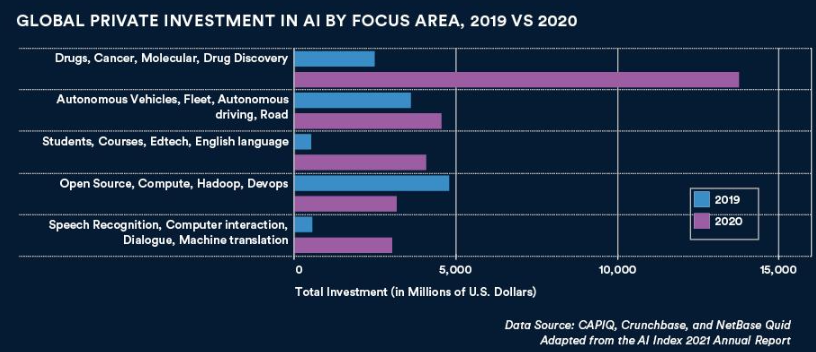
The biggest vertical oppty for AI is in pharma, with more than $13.8 billion in private AI investment — 4.5x higher than 2019. (Stanford, State of AI 2021)
This is already playing out. Today, highly-trained drug researchers spend hours analyzing microscope images — essentially one by one. It’s a painfully repetitive process that creates bias and error, and frankly speaking, I don’t know of very many researchers who enjoy that part of their work! Imagine if you could benefit from the collective bio image analysis work done by every other scientist around the world? This is one application of AI I’m excited for the broader scientific community to have, and it was one of the reasons we invested in Biodock, an AI software platform that automates months of image analysis in minutes.
I’m also excited for AI to empower humans at each step in the value chain — from the discovery to development to distribution of new medicines.
Where are you most excited about the applications of AI in Bio, Sajith? How can biopharma executives harness the latest technologies and, importantly, actually implement them in their organizations?
Sajith: With our customers, there are two main areas where we see AI being directed in the research space:
Target ID and validation: This is about first finding the target. You can think of this as using AI to have a confident understanding of the underlying biological basis of the disease. This relies heavily on techniques like high content screening paired with machine learning.
De novo molecular design: Second is then about designing a drug for that target. As an example, R&D teams are using AI models to actually design a protein that has never been seen before. This relies heavily on large scale, real-world data sets.
With both of these categories, the common challenge we observe is that while many biotech companies aspire to adopt ML, few have the ML-ready data and digital infrastructure required to extract clean, structured, harmonized datasets for training machine learning models at scale.
Biotech organizations need to start with a solid data and digital infrastructure to enable their ambitions here. Universal access to high-quality data at scale has to be the cornerstone to a successful AI and ML strategy. With Benchling, we start by making it easy to capture structured data — scientists are then empowered to ask questions and uncover new insights. From there, we layer in built-in analytics as well as connectivity to each organization’s data science and machine learning efforts, shrinking the gap between wet lab and dry lab.
The industry is starting to see real progress with AI-focused drug discovery companies reaching meaningful milestones or readouts of clinical trials — look at Relay Therapeutics and Recursion. These successful readouts are catalyzing more investment into the space – big pharma has done $20B of AI-based drug discovery deal making in the last 12 months alone, spending hundreds of millions upfront.
On building bridges between technologists and scientists
Jay: As they continue modernizing their enterprises with software, biotech needs to attract, hire, and retain world-class data scientists, ML experts, and software engineers. I appreciate that Benchling is a unique 50/50 blend of people with scientific backgrounds alongside technologists and operators from many other industries. How do you at Benchling build a culture that unites both tech and science? What’s your advice to biotech on how to attract more technologists?
Sajith: Technologists are waking up to the fact that biotech offers the most interesting data and software challenges of this century. Engineers, data scientists, and programmers want to work on groundbreaking innovations with potential for real-world impact, and biotech is an industry that’s curing disease, combating climate change, and eliminating hunger. It’s also an industry where technologists have recently made massive contributions, whether it’s getting COVID vaccines to market faster or making unprecedented advancements in predictive AI, like Chroma from Generate Biomedicines. This mission and overall value proposition is where biotech shines and must stand out with recruiting.
Knowing that recruiting top talent is always a priority, part of this is also about confronting the perceived and real cultural challenges of technologists and scientists working together. To join an organization, people want to know they’ll be able to make an impact, be set up for success, and enjoy the working environment. For a software engineer joining a biotech, they’ll rightfully be curious about whether the environment will be a good fit.
Biotech organizations need to proactively build bridges that unite technologists and scientists. For starters, don’t expect your engineers to know all the science. Help them get comfortable with it. More often than not, an engineer is hungry and curious to learn and will be more confident and productive in your org if you teach them about your science. Once hired, we give people a biology 101 at Benchling. We do this in our onboarding, where all new hires take a biology course we designed. In this way, you help new hires who don’t have a science background feel grounded in the fundamentals and able to use a shared vocabulary with their colleagues.
Likewise, research scientists aren’t going to automatically know how to collaborate effectively with data scientists or software engineers. Leaders at the org must be intentional about how these groups work together, helping scientists to understand at which stage to bring a technologist in to help build a new R&D model, and how to work with their tech partners to ask better questions of the data. Data scientists in particular excel in working with unstructured data sets, transforming this into structure, models, and meaning — their value in an R&D lab is massive. If these teams grow up working independently or in silos, you’re not going to reap the full rewards. You need to build real teamwork and collaboration between technologists and research scientists from the start.
The biotech revolution
Jay: Final question, what about this space interested you so much to embark on the journey of creating a company?
Saji: My background is a mix of both software and science. I grew up programming and studied computer science, but got excited by the possibilities and impact of biology. At university, I really wanted to pursue a career in biotech. It was the contrast between working in science and software that led to Benchling. In my life as a software engineer, I had powerful, rapidly evolving, user-friendly tools that helped me work seamlessly. We could build apps with friends and bring them to the world overnight. In software, the default is collaboration. In contrast, in the biology lab, where I was doing arguably more impactful and complex work, all we had at our disposal were Excel spreadsheets and paper notebooks. Working together was hard. Software might have been eating the rest of the world, but it had failed science.
In that problem, we saw an opportunity: to build software that could not only keep up with the new speed of science but accelerate it.
Biotech can and will be the biggest technology revolution since the internet in terms of impact, scale, and innovation. And I believe that the past decades of progress in the tech sector — particularly the amazing tools for productivity and collaboration — have a place in biotech.
Jay and the team at Andreessen Horowitz would agree, we’re in the early days of bringing tech and biotech together. What’s remarkable about biotech is that all of this innovation will be generated in pursuit of improving lives — making them less painful and more prosperous. That won’t just benefit a few investors in Silicon Valley or the scientists and biotech companies spearheading important progress. It will benefit the entire world.
The views expressed here are those of the individual AH Capital Management, L.L.C. (“a16z”) personnel quoted and are not the views of a16z or its affiliates. Certain information contained in here has been obtained from third-party sources, including from portfolio companies of funds managed by a16z. While taken from sources believed to be reliable, a16z has not independently verified such information and makes no representations about the enduring accuracy of the information or its appropriateness for a given situation. In addition, this content may include third-party advertisements; a16z has not reviewed such advertisements and does not endorse any advertising content contained therein.
This content is provided for informational purposes only, and should not be relied upon as legal, business, investment, or tax advice. You should consult your own advisers as to those matters. References to any securities or digital assets are for illustrative purposes only, and do not constitute an investment recommendation or offer to provide investment advisory services. Furthermore, this content is not directed at nor intended for use by any investors or prospective investors, and may not under any circumstances be relied upon when making a decision to invest in any fund managed by a16z. (An offering to invest in an a16z fund will be made only by the private placement memorandum, subscription agreement, and other relevant documentation of any such fund and should be read in their entirety.) Any investments or portfolio companies mentioned, referred to, or described are not representative of all investments in vehicles managed by a16z, and there can be no assurance that the investments will be profitable or that other investments made in the future will have similar characteristics or results. A list of investments made by funds managed by Andreessen Horowitz (excluding investments for which the issuer has not provided permission for a16z to disclose publicly as well as unannounced investments in publicly traded digital assets) is available at https://a16z.com/investments/.
Charts and graphs provided within are for informational purposes solely and should not be relied upon when making any investment decision. Past performance is not indicative of future results. The content speaks only as of the date indicated. Any projections, estimates, forecasts, targets, prospects, and/or opinions expressed in these materials are subject to change without notice and may differ or be contrary to opinions expressed by others. Please see https://a16z.com/disclosures for additional important information.
Powering breakthroughs for over 1,300 biotechnology companies, from startups to Fortune 500s



