Biotech cloud computing software keeps workflows moving
Slow and steady isn’t going to win the biotech race. In order to stay one step ahead of the competition, biotech companies need the computational horsepower only cloud computing can deliver. Over the last 20 years, advancements in cloud computing have made it a technological advantage for growing biotech companies as they move from research to development and beyond.
Biotech cloud computing helps companies ensure productivity, quality and compliance
Biotechnology companies generate huge amounts of data that need to be recorded, analyzed, and stored in order to advance discoveries through the R&D pipeline, improve productivity, and secure data. Digital solutions are the obvious path forward, but not all software is created equal.
Whether it’s implementing custom builds or system updates, it can take months, even years, to make changes to on-premise software tools due to complex code, poor user interfaces, and the necessity to involve vendors when making changes. What’s more, on-premise software tools often lack flexibility, limiting configuration capabilities to certain groups, applications, and use cases.
Overall, on-premise software tools are unable to keep up with the rapid, exponential data generation in biotech. They’re also unable to offer seamless connectivity throughout the full R&D workflow. This makes companies vulnerable to delays, bottlenecks, and inconsistencies in process execution and data capture. In turn, growing companies must then devote precious time to suboptimal iterations and troubleshooting.
However, all of this is completely avoidable with the right informatics solution. With high-performance cloud computing platforms, like Benchling, you can get around these barriers to keep productivity high, the quality of products consistent, and your workflows and processes compliant.
Here’s how:
Keep your data and your teams connected
Many on-premise software systems are disjointed, creating scattered data silos between departments and teams. This is because they either cannot integrate well with other systems and tools, or they require complex and time-consuming configurations. This makes it difficult for teams to find and share the data and information they need to move projects forward. These problems hinder an organization’s ability to spread valuable information across the company that could make all the difference to a vital program.
Biotech cloud computing platforms seamlessly integrate with instruments and other software to connect your entire workflow and drive full traceability of results. For instance, linking electronic lab notebooks, laboratory information management systems, entity registries, sample inventories, and analysis tools can connect samples to results and provide full experimental history. Scientists can easily access, analyze, and share all the relevant data for a given experiment, ultimately speeding up workflows across the board.
Cloud-based R&D platforms, such as Benchling, provide a centralized data warehouse that serves as a single source of truth, which you can make accessible to any team member despite their geographical locations. Pairing this full connectivity with an intuitive interface and customizable dashboards enables teams to share, analyze, and monitor data efficiently—from discovery through bioprocessing. This way, you can minimize the amount of time lab members spend entering and searching for data, and increase the amount of time they can spend working together to move projects forward.

Keep your lab members connected within and across teams.
Scale and adapt as your organization grows
As companies add new processes, generate more data, and expand teams, they need to be able to view and analyze their entire pipeline and connect the dots between complex data in order to understand project performance, allocate resources effectively, and make data-driven decisions.
The ability of cloud-based solutions to adapt to ever-changing workflows is pivotal to keeping R&D moving forward. Biotech cloud computing software is the only solution flexible enough to let each team use the product however they want, while still maintaining standardization and open lines of communication throughout the overarching, cross-team workflow.
With Benchling, you can customize and adjust workflows, permission hierarchies, entity types, and data fields without a single line of code—so you can make system changes painlessly as your processes evolve and scale.
Learn more about codeless configuration with Benchling
What’s more, biotech cloud computing software like Benchling unifies your workflows and allows you to design and monitor progress in trackable stages. Scientists, managers, and executives can get a 360-degree view of R&D processes and dive deeper into the data in order to:
Optimize R&D output and maintain productivity and reproducibility
Manage R&D processes and analyze the historical outcomes of your programs
Improve how you forecast milestones and resource allocation needs
Rapidly iterate on R&D workflows and manage the complexity of your research to not only create a successful product, but build a successful pipeline of lead candidates
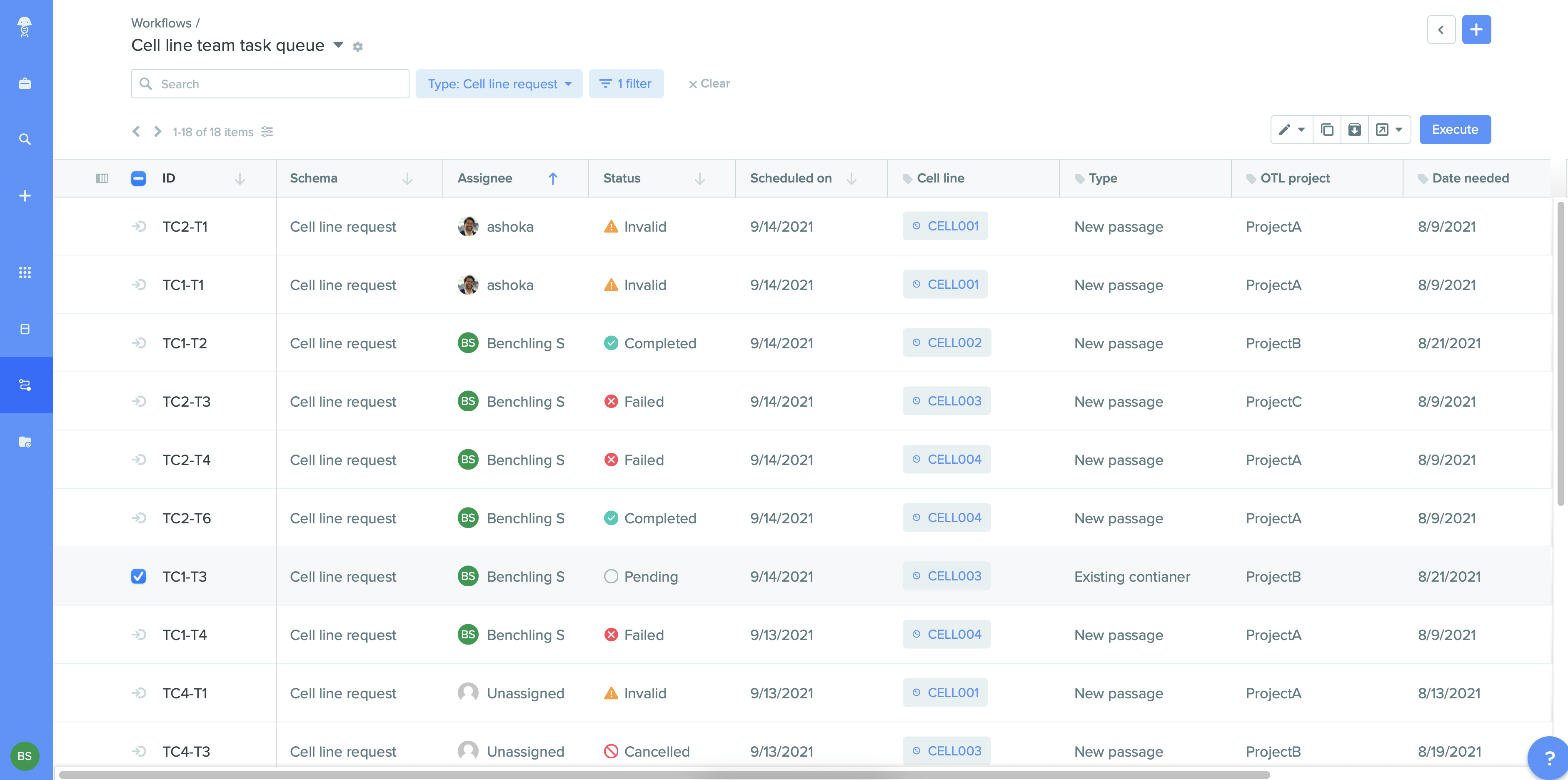
Get a bird’s eye view of your organization’s workflows and optimize for efficiency across processes, teams, and instruments with Benchling Workflows.
Validate workflows for GxP compliance
Biotech cloud computing software has advanced to include the necessary elements to keep end-users compliant with regulatory requirements such as FDA 21 CFR Part 11 and EU Annex 11, including:
Controlled electronic signatures
Audit trails
Entry versioning
User access controls
Visible time stamps
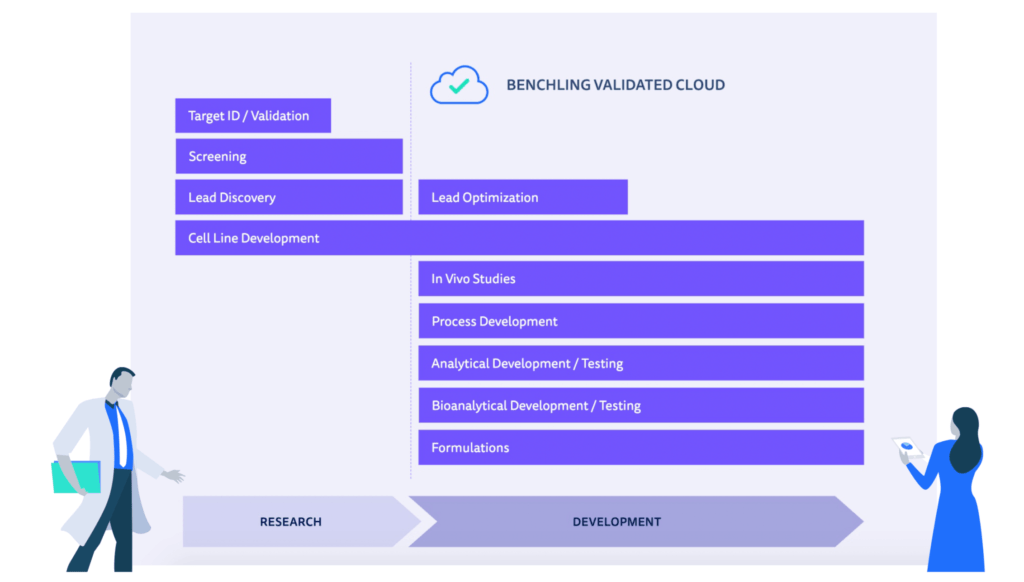
Benchling Validated Cloud supports common lab operations across the R&D life cycle.
With on-premise software, Quality, Regulatory, and IT teams often face laborious, manual processes to validate each new release. In modern cloud computing, software vendors frequently build test scripts that go along with their code, meaning your team can perform thousands of automated regression tests with each new release. Additionally, with multi-tenant cloud computing, companies like Benchling can perform much of the software validation process on behalf of its customers and provide IQ/OQ reports promptly for each release, reducing the overall burden for the company to perform.
With Benchling, R&D organizations have greater control over their workflows, work within a compliant, validated environment, improve tech transfer across their R&D, and can efficiently assemble data packages for regulatory filings.
Get to know Benchling Validated Cloud
Keep your data secure and confidential
In the past, biotech companies have been hesitant to adopt cloud-based platforms because of quality management and confidentiality concerns. A security breach is not only highly disruptive, but can be wildly expensive to reconcile—especially because many companies are working with bread-and-butter intellectual property and, potentially, patient data.
Modern biotech cloud computing software operates under a Quality Management System (QMS) and has an established Information Security Management System (ISMS) to ensure the confidentiality, integrity, and availability of customer data. Further, cloud-based platforms can be equipped with a comprehensive set of admin controls to govern permissions, accessibility (including MFA/SSO), and data management—so only the people who are supposed to see the data get access to it.
Biotech cloud computing with Benchling
Benchling is changing the game with faster implementations, seamless integrations, and stellar adoption rates. Our industry-leading Life Sciences R&D Cloud provides a universal source of truth for the entire R&D lifecycle—built to adapt to the needs of every team. In fact, more than 200,000 biopharma, agritech, and industrial biotech scientists trust Benchling to improve collaboration, increase productivity, and accelerate innovation.
Benchling offers a suite of six unified, cloud-based applications
Benchling’s unified capabilities, codeless configuration, and integration with lab automation instruments allow life science companies to unite every step of R&D on a single platform.
Notebook: Ensure documentation completeness and compliance
Molecular Biology: Accelerate DNA and amino acid design, at scale
Registry: Standardize, connect, and contextualize sample data
Inventory: Track and manage every sample and reagent
Workflows: Design, test, and optimize R&D processes
Insights: Translate R&D data into actionable insights
The average scientist saves more than 1 day per week when they switch to Benchling.
Calculate the impact Benchling can have on your R&D.
Benchling maintains the highest level of quality, security and protection of our customers’ data
Benchling operates under a well established Quality Management System (QMS) that is aligned with ISO 9001 and ICH Q10 guidelines. Benchling is ISO 27001 certified and keeps data secure and protected under the leadership of our chief information security officer (CISO).
Get an overview of the Benchling Validated Cloud and how it ensures traceability, accountability, and data Integrity.
Read the Datasheet for Benchling's Validated Cloud
Benchling ensures compliance through the Validated Cloud
Benchling Validated Cloud provides a solution for using the Benchling Life Sciences R&D Cloud in validated environments. Benchling applications, such as Notebook, Registry, and Inventory, are deployed in a dedicated tenant with controlled product releases, which helps R&D, IT, and Quality teams maintain Benchling as part of their validated (GxP) workflows.
What’s more, with Benchling, you can use validated tenants that are separate and distinct from non-validated tenants to carefully and predictably control the product release process. This is particularly advantageous in biotech R&D where scientific discovery teams prefer flexible data systems for iterative experimentation and development teams require control, reproducibility, and scalability for high-quality manufacturing and adherence to stringent regulatory guidelines.
Learn more about how a modern digital infrastructure is crucial to addressing a new set of challenges that come with the next generation of therapeutic development.
Powering breakthroughs for over 1,300 biotechnology companies, from startups to Fortune 500s
